Is Dengue really controlled using Wolbachia infected mosquitoes in Colombia? (NM3-8eng)
Making predictions is difficult, especially about the future
As my spanish readers know, I started this newsletter to offer a critical appraisal of the old and new methods to control dengue outbreaks, especially aiming at the public health agents and politicians in Argentina. I covered the use of Wolbachia infected Ae. aegypti in the post NM3.4. After going through the details of several publications about the successful advances of the program according to the World Mosquito Program, and about the critical observations published in scientific journals and professional networks, I considered appropriate to make another revision of the subject. Critical observations include the loss of infection by Wolbachia due to a number of causes (especially high temperature, example of scientific articles here and here), doubts about the impact of the method on dengue outbreaks, loss of capacity of the Wolbachia infected mosquitos to block virus transmission. (example of scientific article here), among others.
In one of the last publications, the World Mosquito Program claims that “Since the large-scale roll-out of Wolbachia-infected Ae. aegypti mosquitoes across a continuous population of 3.3 million people in Bello, Medellı́n and Itagüı́, Colombia, the incidence of notified dengue cases has been 95–97% lower than during the decade prior to Wolbachia introduction” (extracted from the 3 first lines of the Discussion section of the PLOS-NTD 2023 paper “Reduced dengue incidence following city-wide wMel Wolbachia mosquito releases throughout three Colombian cities: Interrupted time series analysis and a prospective case-control study” (link to the article here). As the claim is quite spectacular, and several health agencies (in Argentina and elsewhere) are considering the adoption of the Wolbachia method, I took a close look to the PLOS-NTD paper. This post will tell you what I found.
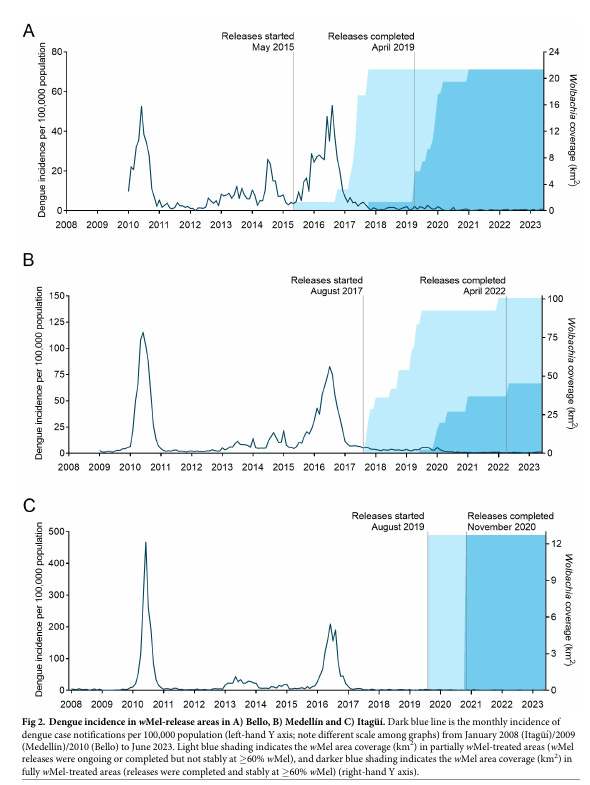
The claim that the incidence of notified dengue cases has been 95–97% lower is basically supported by the variation of dengue incidence from 2008 to 2023, as informed by the Instituto Nacional de Salud and made available by the authors in this link. The data basically look like the following figure (taken from mentioned the PLOS-NTD 2023 publication)
Blue and light blue shades show the period during which the release of Wolbachia infected Ae. aegypti (Wolbitos from now on) occurred. As the authors kindly provided the original data of dengue incidence in the 3 localities of the Aburrá Valley, I aggregated the data for the 3 localities and made a similar figure as follows
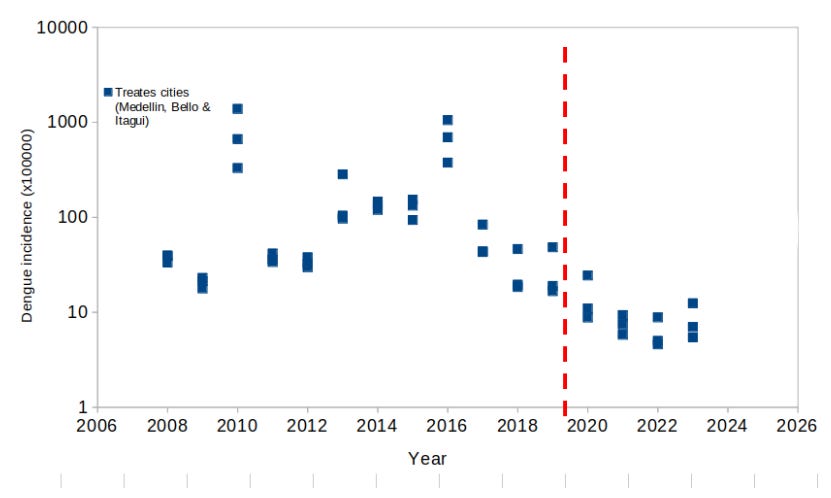
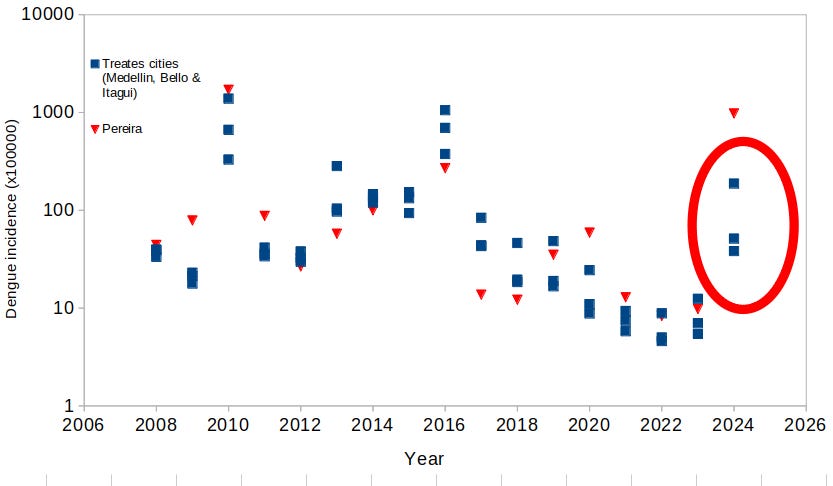
The blue squares show the dengue incidence per year in the treated localities of Bello, Medellin and Itagui (note the logarithmic scale). The vertical dashed red line show the moment when the release of Wolbitos started. Authors compare the dengue incidence after the release against the dengue incidence before the release. Clearly this gives the 96% dengue reduction mentioned by the authors.
Dengue epidemiology is a bit tricky, as it shows outbreaks followed by periods with few cases. We will not discuss the causes of the behavior of the disease here. Periods between outbreaks varies between 2-4 years, although longer periods might happen. Additionally, the outbreaks and the few-cases-period do not occur at the same time everywhere, for reasons we do not understand completely, or at least are too complex to measure. Because of this temporal and spatial heterogeneity of dengue incidence I wondered about the situation in cities near the treated area of the Aburrá Valley. Fortunately, the PLOS-NTD 2023 authors pointed to the file where the Instituto Nacional de Salud Pública of Colombia shows the dengue incidence in the 57 cities with more than 100.000 inhabitants (link here). Within these cities, Pereira is located 250 kilometers south of Itagui, where dengue incidence appears as follows:
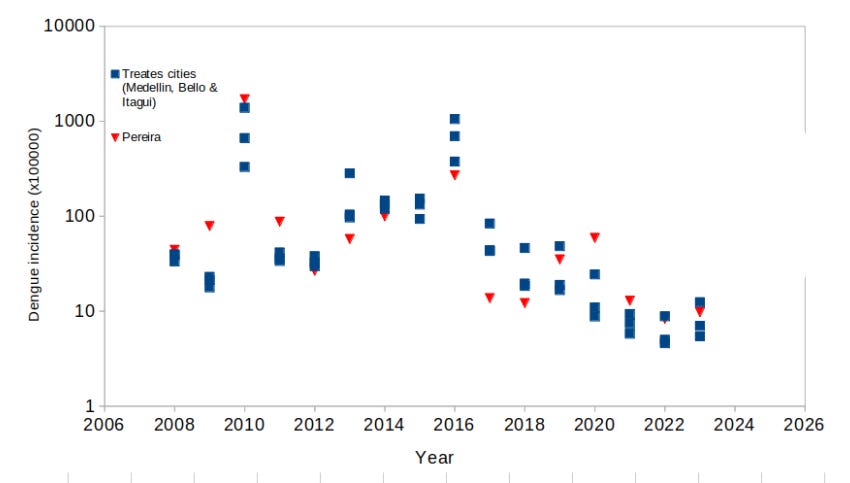
Same figure as before, but superimposed as red triangles the dengue incidence in the city of Pereira from 2008 to 2023. As far as we know, Pereira was not included as an area where the Wolbitos were released; however, the dengue incidence resemble very closely to what happened with the incidence in the treated area of the Aburrá Valley. Unless there was a very unlikely migration of Wolbitos 250 kilometers south, there is something difficult to understand about the differential effect of the Wolbitos. Did the dengue incidence decrease everywhere (or at least in some places) by pure chance? Maybe, but we don’t have data to explain the cause.
As 2024 was the worst year for dengue outbreak in South America, I looked at the dengue incidence of 2024 in the 3 treated cities of the Aburrá Valley, as published by the Boletín Epidemiológico Semanal (week 50) of the Instituto Nacional de Salud of Colombia (link here). Although we would expect a flat line for the incidence in the 3 treated cities, it climbed as seen in the figure below.
Same figure as before, with the addition of the dengue incidence in Medellin, Bello and Itagui (3 blue squares, enclosed in the red ellipse) and Pereira (red triangle) for the year 2024. So, these data begs for questioning: is this showing the Wolbitos were not able to stop the outbreak? Did they block a much worse outbreak? We don’t have data to answer, but shouldn’t we expect for the Aburrá Valley a number of cases like the ones right after the Wolbitos release in 2019? The spreadsheet I used can be downloaded here.
With these data in mind, I looked at another piece of the data-rich article of PLOS-NTD 2023: the wMel infection prevalence in local Aedes aegypti mosquito populations per month along the whole study period (figure link here and included below for convenience).
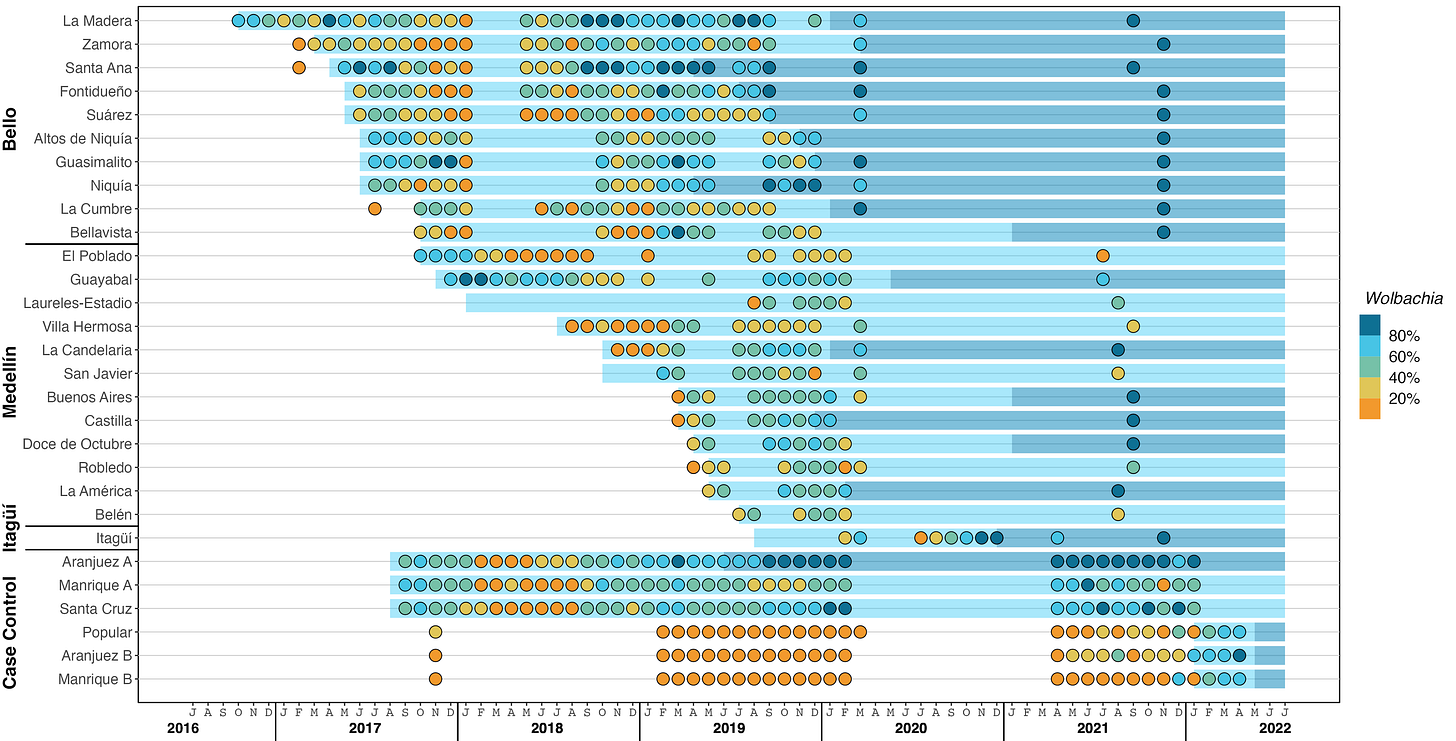
According to the Wolbachia method, after the release of Wolbitos during 12 to 20 weeks, the local population of Ae. aegypti would be replaced by the released Wolbitos, at least representing 60% of the mosquitoes flying around the area.
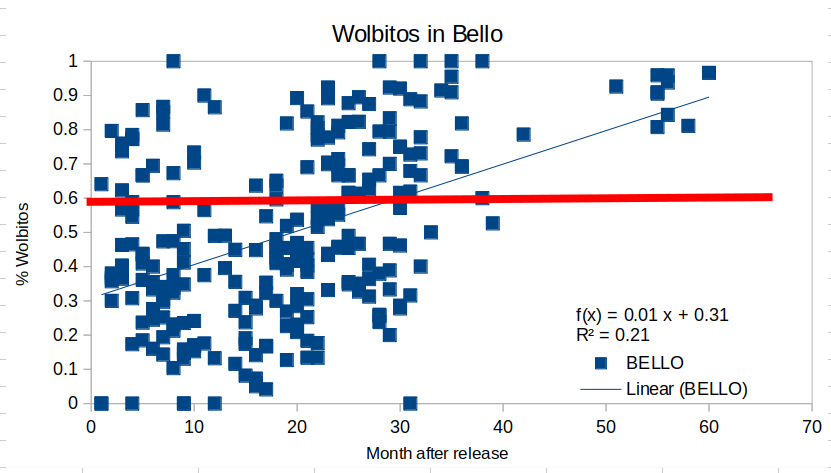
A much more detailed database is included in the data availability statement of the article, as a convenient spreadsheet (link here), containing the analyzed number of Ae. aegypti field collected eggs, and the number of these eggs infected by wMel for each neighborhood and month, with indication of the moment when the Wolbitos releases began in the neighborhood. I used the data on the spreadsheet to make a traditional dispersion graph (without the aggregation that the authors used to make the categories of the above figure) to show the fraction of field collected eggs of Ae. aegypti infected by wMel against the time (in months) since the first release. I did two graphs one for Bello and another for Medellin. The graph for Bello resulted in
Each blue square is the fraction of field collected eggs infected by wMel along the time since the first release (0 in the X axis). The thin blue line is just a linear trend of the data set and the thick red line at 0.6 means (according to the authors) that below that line, Wolbitos must be released to maintain a minimum of 60% Ae. aegypti population infected by Wolbachia. Over the 0.6 line, release of Wolbitos can be stopped. First thing that called my attention was the wide dispersion of the data, except the few data collected after 50 months of the first release. The second interesting feature is that the linear trend is positive, but on average it took 30 months to cross the 0.6 line of “stop-releases”, quite a few time more than the one mentioned in the WMP web page where it says “The release period usually lasts for 12 to 20 weeks” (link here), about 6 times the figure actually.
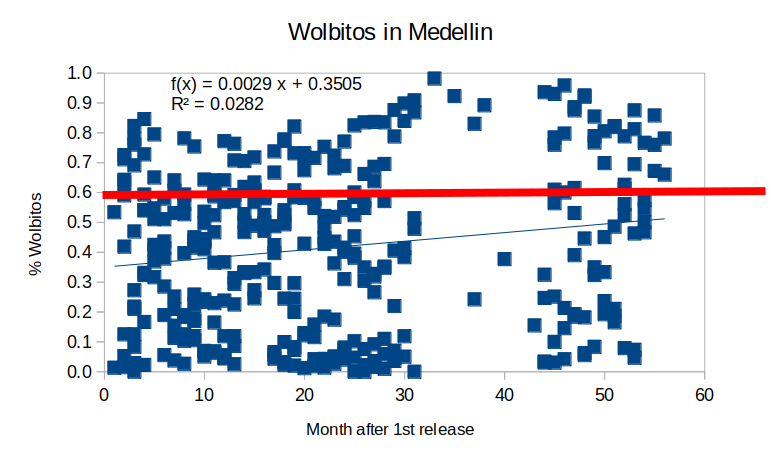
When I did the same graph for Medellin I’ve got
In this case, the dispersion is more dramatic, and although the trend is positive, it is clear that it is not different from 0. And even more interesting is that taking the trend as the average infection by wMel since the first release, it never reaches the 0.6 “stop-release” line, meaning that you should keep releasing Wolbitos after more than 50 months after the first release. The spreadsheet I used to make these graphs can be downloaded here.
So, what is the message here? Although I am a fan of the scientific knowledge use for the control of disease vectors, I always keep an eye on two things: do no harm, and do not waste public money. The Wolbachia method is an exquisite example of the attempt to use science to improve the well being of people. The beauty of the method for the advance of science-based knowledge (as contrasted with the faith-based knowledge), is that it is always under the scrutiny of fellow researchers. In the case of the dengue control in the Aburrá Valley, a couple of concerns appear: a) is the variation of dengue incidence driven by the Wolbitos or by the intrinsic epidemiology of the disease that has nothing to do with the Wolbitos? b) does the released Wolbitos really replace sustainably the local population of Ae. aegypti in every attempted case or is it a case of “depends”, and sometime it works and sometime it does not? Are we expecting too much from this technology? Are we seeing a repetition of the fallacy of the Concorde? (we invested too much to stop now). It seems that, at least in the case of Colombia, independent data is needed to verify the claims about the impact of the technology over the dengue outbreaks. As always in science it’s a matter of time. The truth will prevail in the end.
Thank you for your interest in the newsletter No Me Molestes Mosquito. You can subscribe using the button below, and you can share this with your contacts.


Controlling the dengue vector is a difficult task. There is not enough space or time to elucidate the issue as it deserves, but dengue was eliminated from much of the continent long before Wolbachia, cytoplasmic incompatibility, or any other tongue twister appeared.
The problem we have is simple: the vector control programs against dengue and malaria – and even those for Chagas disease – were eliminated shortly after declaring victory against the disease as they were considered unnecessary. More tragically, many do not understand or want to accept that, like antibiotics in medicine, "anti-mosquito" materials are medicines for the environment. As such, they have to be used when necessary and following specific protocols. Moreover, few in medicine seem to understand this or the concept of integrated vector management. As with antibiotics, the use, abuse and almost exclusive dependence on "drugs of the moment" tends to confuse a population that does not understand the reason for their use, considers them toxic materials, and often results in the development of tolerance or resistance to them by the vector. And let's not forget that many of these conditions are cyclical and affected by changes in climate.
The situation in Colombia is interesting. I don't think it's a "cause and effect" directly linked to the use of Wolbitos, as we don't have details of how the plan was designed or implemented. In short, we must return to integrated vector management combined with the active participation of communities to make them less favorable to breed their own mosquitoes.
Manuel Lluberas MS IDHA